Rock University High School CIF Mini-Project 2018
Important Discoveries
|
In 1911, a physicist from New Zealand named Ernest Rutherford discovered the nucleus. Rutherford also proposed a name for the positively charged part of the atom in 1920, and proposed there was a neutral particle in the nucleus, and in 1932, a student of Rutherford and British physicist James Chadwick confirmed that theory. Rutherford discovered positively charged particles in an experiment with cathode-ray tubes. In 1897, a British physicist, J.J. Thomson discovered the electron. In December of 1938 fission was discovered on accident by Otto Hahn and Fritz Strassmann. Fusion was first thought of in Cambridge, UK during the 1930’s, but was abandoned, and then revisited after World War II. The Manhattan project was a very big pushing factor for the possibility of nuclear fusion. Hans Bethe, a German born physicist first recognized that fusion of hydrogen nuclei is also used by the stars. Bathe won a nobel prize later on for work he did contributing to the research.
|
Basic Components
Nuclear weapons release a large amount of energy in a small amount of time by a chemical reaction. There is two main types, fission and fusion, both of which use atoms. Atom is the smallest particle of matter. Fission is when atoms are split apart, used to make atom bombs, Fusion is when atoms are are fused together, used to make hydrogen bombs. Fission Bombs are normally made with a core of plutonium or highly enriched uranium, the only materials that can be used to achieve a self-sustaining chain reaction. There are two ways to make fission happen, the gun method, and the implosion method. In the gun method, used for the Hiroshima bomb, there uranium that is fired down a barrel into more uranium. The implosion method is far more efficient than the gun method. The implosion method works when you surround a sphere of plutonium or uranium with explosives that all go off at once. Fusion bombs, however, used deuterium and tritium fused together to make heavier atoms. Fusions bombs are 10 to 100 times more explosive as those that were dropped on Hiroshima and Nagasaki.
Physical Impact
The destruction that the blasts from these weapons depends on how close things are to it. You can still see the shadows of the people who were vaporized from the bombs dropped on Hiroshima and Nagasaki. There is also radiation from bombs. Thermal radiation is light and heat, which can be seen, turn people blind, and explode sand and flammable materials can catch fire from miles and miles away. Effects of radiation on the humans can be measured by severity, and each level of severity is measured by rads, which is 100 ergs of energy per gram of tissue. There are 4 levels of doses, Extremely high, high, moderate, and low. |
|
Diving Further into the Science
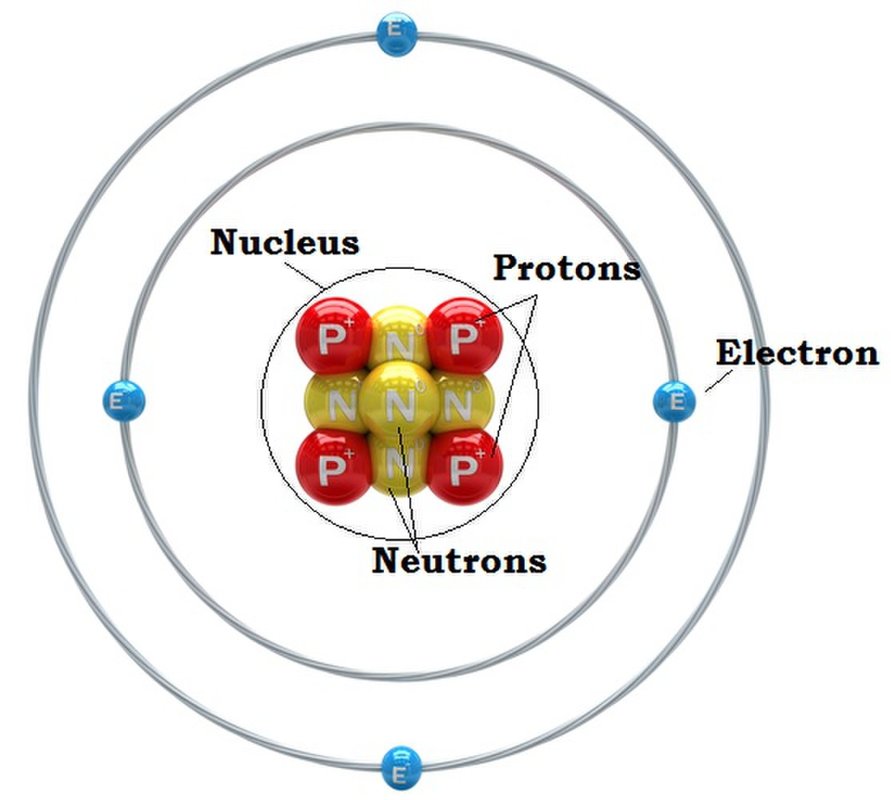
The Atom
|
Atoms are the smallest and most basic units of matter. Atoms are made up of three different parts: neutrons, protons, electrons. Within the nucleus, the center of the atom, protons and neutrons are clumped together. Neutrons have a neutral charge and protons have a positive charge. Neutrons and protons make up most of the mass of an atom. The number of protons determines what element something is. The number of protons is also called the "Atomic Number" on periodic tables. Adding or taking away neutrons creates an isotope. Electrons have a negative charge and travel in a cloud that orbits the nucleus.
Fun Fact: The word for atom was derived from the Greek word for "indivisible." |
Radiation and Critical Mass
|
An element is radioactive when the nucleus is unstable. This can be due to an imbalance of protons and neutrons. Ideally the number of protons and neutrons would be around the same. Radioactive decay is when the unstable atoms start giving off particles to become stable.
Also, there is a chance that one fission event won't trigger another one because we cannot control what direction a neutron flies out nor where it goes. Critical Mass is defined as the quantity of a particular product, on average, needed to possibly create another fission event through neutrons being sent out in different directions. |
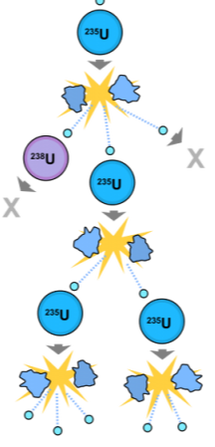
Fission
|
Nuclear fission is when an atom is split apart due to the nucleus being collided with a neutron at high speeds. This causes different fragments to fly out at high speeds. These fragments include different elements and neutrons. The neutrons that are flying out can then hit the nucleus of another atom which causes a neutron to fly fast into another nucleus. This phenomenon is referred to as a chain reaction. For a proper chain reaction to occur, you have to have enough of the given radioactive element because there is a slight chance it wont always hit it's mark. Fission produces less energy than fusion.
|
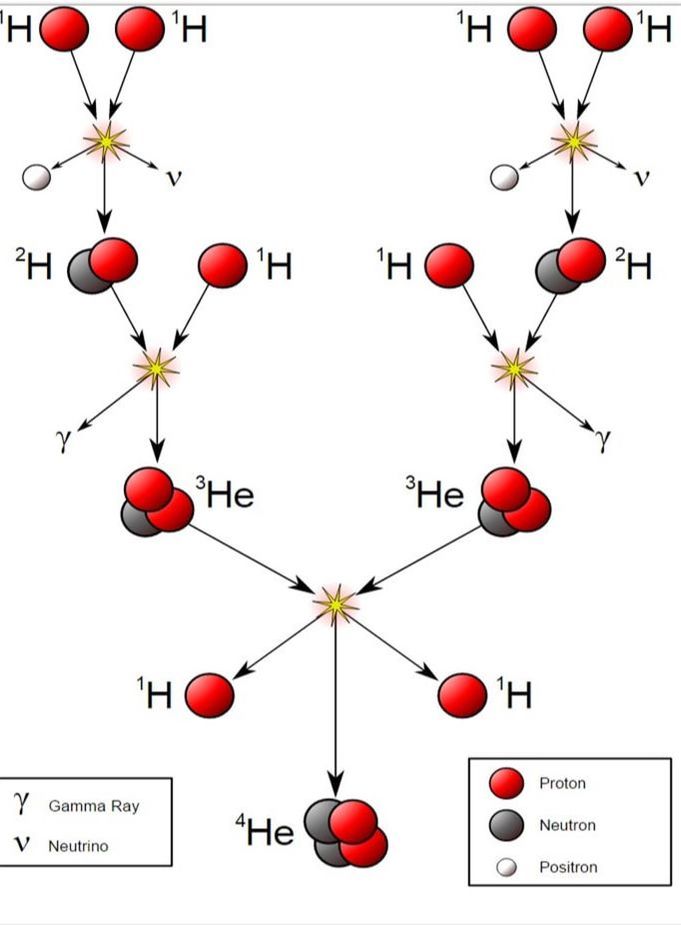
Fusion
|
Nuclear fusion is when nuclei of different atoms collide at high speeds, combining to create one or more different nuclei, energy, and other subatomic particles. It's important to note that fusion creates a lot more energy than fission. Nuclear fusion is also what powers the sun and many other stars. On stars like the sun, the chain reaction is seemingly unending.
|
Behind the First Nuclear Explosion Test, "Trinity"
|
On July 16th, 1945, the first ever nuclear explosion occurred in New Mexico. The Trinity test involved a device called Gadget. The reason they wanted to test it is because they were not quite sure what would happen, some of the guards were even worried it could destroy the whole planet. The bomb was an impulsion type mechanism in contrast to the gun type mechanism which they were positive would work. The Gadget had the same explosion design as "Fat Man," the bomb dropped on Nagasaki. "Little Boy," the bomb dropped on Hiroshima was a gun-type bomb. Both Gadget and "Fat Man" used the isotope plutonium-240. The first layer of the bomb’s core was made of an explosive lens. The explosives would go off and press the central sphere together and charge the uranium. The pressure created by the explosions made the plutonium reach critical mass, which caused it to give off electrons. The isotope uranium-235 used was then ran into by the neutron and split into two atoms. In this case, krypton and barium were made upon the process of fission. The split then creates high amounts of energy. The reaction keeps going on until there is no longer and uranium left, because the fission process creates two more neutrons which continues to split uranium atoms in a chain reaction.
|
|
Photo used under Creative Commons from Greenwich Photography